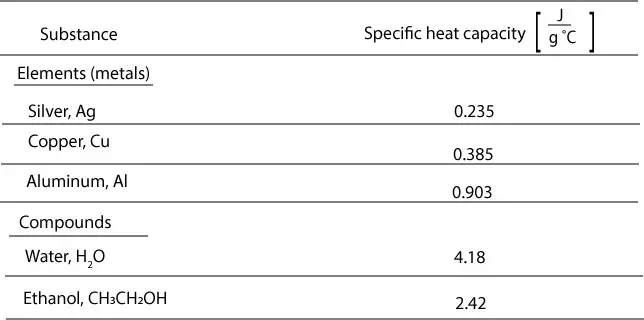
I would like to share my personal exploration of this intriguing problem. What is it about the teaching of this topic or the question that produced this result? Having encountered this type of calorimetry problem when I was a college professor, I reflected on how I approached this question and it made me even more curious about the result. It was a coffee cup calorimeter question that only 15% of the students answered correctly (difficulty 0.15) and there was no correlation to overall success on the test (discriminating level 0.02). What is the ΔHsol'n for the dissolution reaction below? Assume the specific heat capacity of the solution approximates that of water at 4.184 J g-1 C-1 and the calorimeter has 100% efficiency. The final temperature of the calorimeter contents is 34.6 C. Question 14: A student places 0.750 g of solid sodium hydroxide (NaOH) into 20.00 mL of water at 25.0 C inside a coffee cup calorimeter. 
She analyzed what it was about the question on the CCC that produced such a poor discriminating index. She challenged us to look more critically at all of our tests and evaluations and ask whether they evaluate what we think they are evaluating. 
In her article “2016 Canadian Chemistry Contest: Worst discriminators” (March 2017, Chem 13 News) Jennifer Pitt-Lainsbury described her analysis of a CCC question with a low discriminating index and asked for comments (see boxes, below).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
